|
The general trend here is that all of these, these are very high in metallic nature, and these are very low metallic nature which also makes you probably realize while most of the periodic table is a. German chemists Ida Tacke and Walter Noddack set out to find the elements, a quest being pursued by scientists around the world. Pure calcium, actually, this is actually a picture of pure calcium. Mendeleev had predicted, and others including Henry Moseley had later confirmed, that there should be elements below Manganese in Group 7. Not to miss out, element 102, Nobelium, No, is named in honor of Alfred Nobel, who set aside his vast fortune to establish Nobel Prizes.\): Elements in the periodic table are organized according to their properties.Įven after the periodic nature of elements and the table itself were widely accepted, gaps remained. Mendeleev never received a Nobel Prize for his work, but element 101 was named Mendelevium, Md, after him. All the alkali metals are found in nature, but being highly reactive, they do not occur freely in their pure form 1. For instance they discovered phosphorus when they isolated it from urine. The alkali metals, also known as the alkali metal family is a group of six elements characterized by common physical and chemical properties, a similar electron configuration, and shared periodic trends. Over time these gaps have gradually been filled in as scientists unearthed new elements. A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column ( Figure 2.26 ). Not only did Mendeleev arrange the elements in the correct way, but he also had the foresight to leave gaps for undiscovered elements. In the typical periodic table, each element is listed by its element symbol and atomic number. The periodic table only lists chemical elements, and it includes each isotope of each element within one cell. At that time, he had only 50 elements to arrange. The periodic table is structured as an 18 X 7 grid positioned above a smaller double row of elements. He wrote the properties of the elements on pieces of card and rearranged them until he realised that, by putting them in order of increasing atomic weight, certain properties of elements regularly occurred. An interactive Periodic table can be found Periodic Table of the Elements, LibreTexts. The semimetals lie along a diagonal line separating the metals and nonmetals. The metals are on the bottom left in the periodic table, and the nonmetals are at the top right. Then in 1869, a Russian scientist called Dmitri Mendeleev produced one of the first practical periodic tables. 1: The Periodic Table Showing the Elements in Order of Increasing Z. Using Cannizzaros atomic weights, Mendeleev and Lothar Meyer made a great discovery, the periodic law: If. aluminum oxide, Al 2 O 3: aluminum has a valence of 3. hydrogen sulfide, H 2 S: sulfur has a valence of 2. 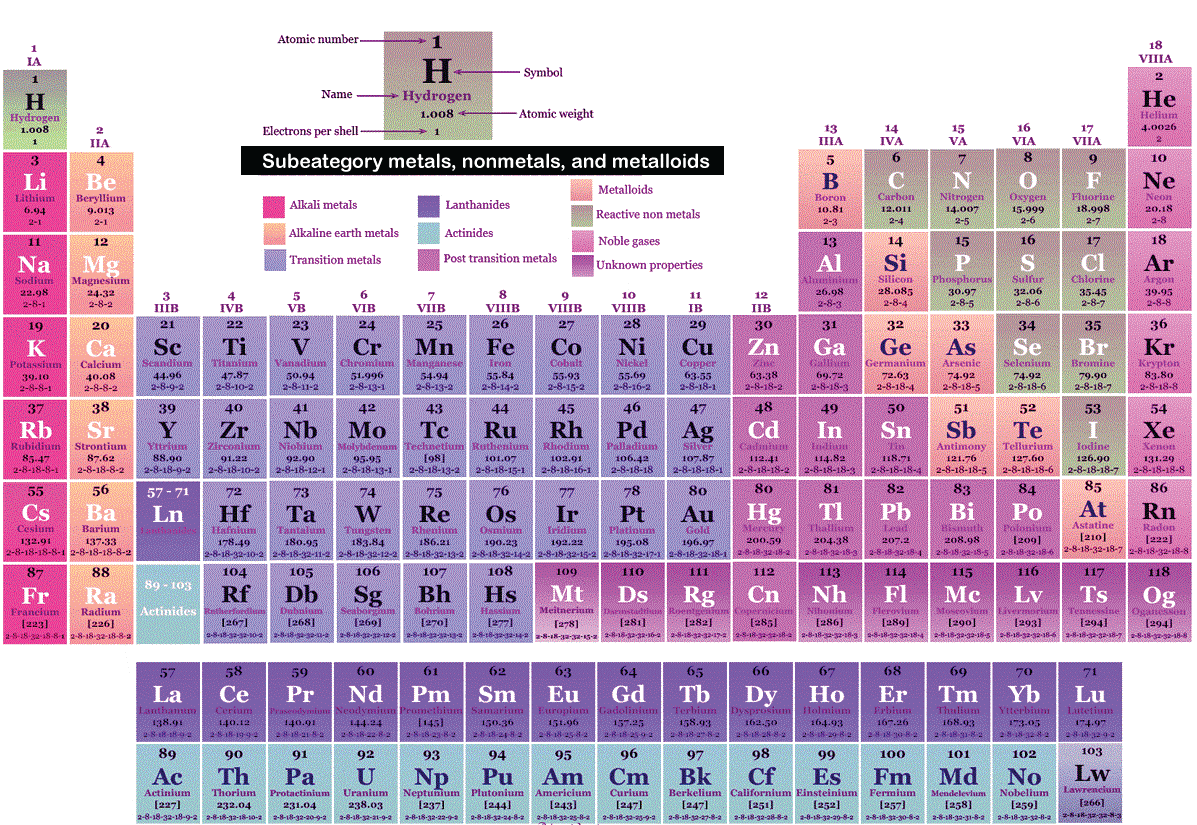
Several other attempts were made to group elements together over the coming decades. lithium oxide, Li 2 O: lithium has a valence of 1. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths. Group 3-12: Transition and Inner transition metals group. Group 1: Alkali metals group (hydrogen not included) Group 2: Alkaline earth metals group. Even after the periodic nature of elements and the table itself were widely accepted, gaps remained. There are total 18 different groups in Periodic table. 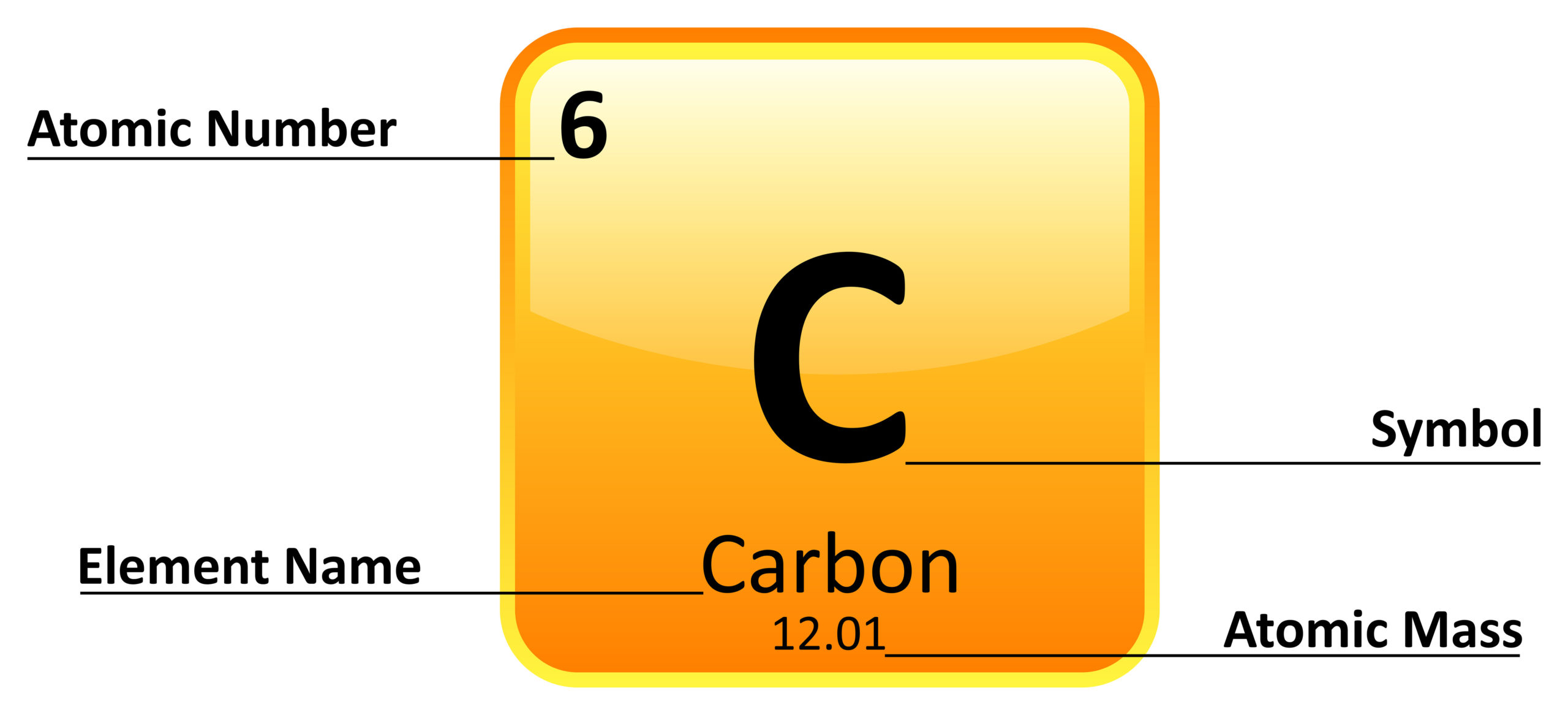 
2: Elements in the periodic table are organized according to their properties. The discovery of other elements followed regularly and soon it became necessary to arrange them in some sort of order. A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column (Figure 3.2b). For the table to fit on a single page, parts of two of the rows, a total of 14 columns, are usually written below the main body of the table. The term is also spelled ionisation energy (British English). By definition, ionization energy is the minimum energy needed to remove the most loosely bound electron from a gaseous atom or ion. However, the first scientific discovery of an element occurred in 1649 when Hennig Brand discovered phosphorous. It is a periodic table trend that increases moving across the table and decreases moving down it. Copper has been used by humans for as much as 7000 years and elements such as gold, silver, tin, lead and mercury have been known for many thousands of years.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
